There’s a push in the UK, by some industries, to replace methane in the gas grid with hydrogen. Proponents claim it is clean, easy to produce, and CO₂ free, with nothing but clean water vapour released.
Ignoring, for a moment, the economic and practical challenges of producing, transporting, and storing hydrogen at grid scale, let’s entertain the idea. Suppose we did it. Suppose every boiler in the country was burning hydrogen tomorrow — what actually happens?
The fuel is only part of the story
Everyone knows that diesel engines produce NOx emissions, nitrogen oxides. What’s less obvious is where the nitrogen comes from. It’s not the fuel, but the air. When diesel combusts at high temperature, it heats the surrounding air enough to break apart N₂ molecules into individual nitrogen atoms. These atoms are highly reactive, and when they encounter O₂ they readily form nitric oxide (NO), which oxidises further to nitrogen dioxide (NO₂). Together, these are referred to as NOx.
The same thing happens in a gas boiler. Fuel doesn’t burn in pure oxygen. It burns in air, which is roughly 78% nitrogen and 21% oxygen. At moderate temperatures nitrogen is inert and passes through the flame untouched. But above about 1,300°C, the N₂ bonds start breaking apart and the same NOx-forming reactions kick off.
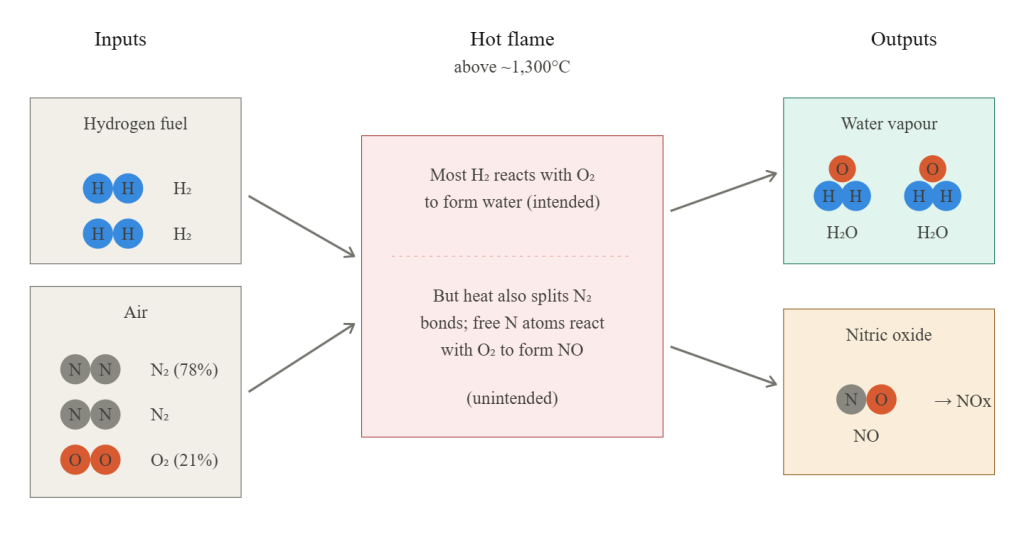
This process is called the Zeldovich mechanism, and the key thing to understand about it is that NOx formation doesn’t increase gradually with temperature. It increases *exponentially*. A small rise in flame temperature produces a disproportionately large rise in NOx. Which brings us to hydrogen.
Why hydrogen releases more NOx than methane
Hydrogen burns *hotter* than methane. The adiabatic flame temperature of hydrogen in air is around 2,100°C, compared to roughly 1,950°C for methane.
That might not sound like much, but because of the exponential relationship between temperature and NOx formation, that 150°C gap translates into significantly more NOx coming out of a hydrogen flame than a methane one.
So why does hydrogen burn hotter than methane in the first place? It comes down to what the released energy has to heat up. When methane (CH₄) burns, each molecule reacts with two O₂ molecules to produce one CO₂ and two H₂O. A lot of the heat released goes into warming up the CO₂, which soaks up energy into its vibrational and rotational modes. Hydrogen combustion does not introduce carbon: 2H₂ + O₂ → 2H₂O. So there is no CO₂ to warm. Instead, the only combustion product is water vapour, so more of the released heat ends up as raw temperature rather than being spread across the products, and consequently results in a higher flame temperature and more NOx formation.
So is hydrogen really zero emission?
It’s true there’s no CO₂ at combustion. But there’s more NOx.
I’m sure manufacturers will engineer workarounds — SCR systems (AdBlue, essentially), exhaust gas recirculation, lean-burn designs. But at that level of added complexity it sort of defeats the purpose of condensing boilers, in that they are cheap to make and install. So it leads to the inevitable: why not just spend the money on nuclear power and heat pumps?
